Regulatory Compliance – FDA & ISO
Achieve compliance excellence with single source asset management software that meets your FDA and ISO regulatory compliance requirements through features like electronic signatures, simple audit trails, and streamlined procedure versioning.
Maintenance Connection Compliance Solution
Maintain compliance with FDA regulations including 21 CFR Part 11, improve production and quality processes, extend asset life, and ensure data integrity.
Regulatory Compliance
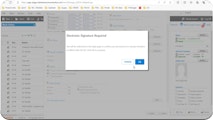
Achieve seamless FDA regulatory compliance with electronic signatures and controlled documentation across all departments.
Enhance Security & Safety
Ensure safety and security, safeguard against errors and unauthorized access, and fortify cyber resilience in alignment with the latest FDA regulations.
Process Efficiency & Automation
Enhance process efficiency and ensure continued compliance with our automated, integrated solutions for seamless electronic signature validation.
Data Trustworthiness
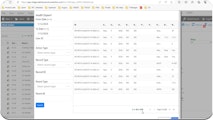
Ensure data trustworthiness and accountability in FDA-regulated activities with transparent, traceable, and verifiable data collection and reporting.
Maintenance Connection Compliance
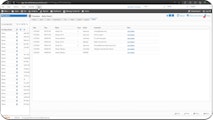
A comprehensive CMMS solution that helps organizations become and stay compliant and analyze, manage, and track their assets throughout all stages of the asset lifecycle.
The Power of Compliance: Signed, Sealed, and Streamlined
In the realm of FDA-regulated industries, compliance isn't just a standard to meet – it's your commitment to excellence. With Maintenance Connection, achieving compliance does not have to come at the expense of decreased operational efficiency and profitability.


.png?u=https%3A%2F%2Flive-accruent.pantheonsite.io%2Fsites%2Fdefault%2Ffiles%2F2024-04%2FIndustry-icons-2022_foodservice_color%2520%25281%2529.png&a=w%3D26%26h%3D24%26fm%3Dpng%26q%3D75&cd=b26b038d55cf088417940f29f8475a26)




